Class 11, Chemistry, Chapter 3,Classification Of Elements & Periodicity In Properties (Solved questions)
Q.3.1: What is the basic theme of organisation in the periodic table?
Table of Contents
ToggleAns: The basic theme of organisation in the Periodic Table is to simplify and systematise the study of physical and chemical properties of all the elements and their millions of compounds. This has made the study simple because the properties of elements are now studied in form of groups rather than individually.
Q.3.2: Which important property did Mendeleev use to classify the elements in his periodic table and did he stick to that?
Ans: Mendeleev used atomic weight as the basis of classification of elements in the Periodic Table. He arranged 63 elements known at that time in the Periodic Table on the basis of the order of their increasing atomic weights and he placed elements with similar properties in same group.
He stuck to this basis sincerely as some places were left vacant for new elements which were not discovered at that time.
For example, Ga (gallium) and Ge (germanium) were not known at that time. He left vacant space for them and named them as eka-aluminium and eka-silicon.
He predicted the existence of these two elements and also described some of their general physical properties.
Later, these elements were discovered and their properties were found to be similar to those predicted by Mendeleev.
However, he also found some elements that did not fit in his scheme of classification on the basis of atomic weight.
In such cases, he ignored the order of atomic weights e.g., iodine is placed after tellurium due to its similarity in properties with other halogens although its atomic weight is lower (=126.9) than that of tellurium (=127.6).
Q.3.3: What is the basic difference in approach between the Mendeleev’s Periodic Law and the Modern Periodic Law?
Ans: Mendeleev periodic law It states that the properties of the elements are a periodic function of their atomic weights.
Modern periodic law It states that the properties of the elements are a periodic function of their atomic numbers.
Thus, change in the base of classification of elements from atomic weight to atomic number is the basic difference between Mendeleev’s periodic law and the modern periodic law.
Q.3.4: On the basis of quantum numbers, justify that the sixth period of the periodic table should have 32 elements.
Ans: In the modem Periodic Table, the 6th period begins with filling of principal quantum number, n =6.
According to Aufbau principle, in the ground state of the atoms, the orbitals are filled in order of their increasing energies. Therefore, in 6th period, e– enter in 6s, 4f, 5d and 6p subshells. Total 16 orbitals (2 + 7 + 5 + 3 respectively) are present in these subshells. According to Pauli’s exclusion principle each orbital can accommodate maximum two e–, therefore 16 orbitals can have 32 electrons and hence, 6th period have 32 elements.
Q.3.5: In terms of period and group where would you locate the element with Z =114?
Ans: Z114 = [Rn]86 7s2, 5f14, 6d10, 7p2
In the Periodic Table, the element with Z = 114 is located in Block – p-block (as last electron enters in p-subshell). Period – 7th (as n = 7 for valence shell) Group – 14th (For p-block elements, group number = 10 + no. of e– in the valence shell)
Q.3.6: Write the atomic number of the element present in the third period and seventeenth group of the periodic table.
Ans: General configuration for 17th group elements is ns2 np5. In the third period, the principal quantum number for valence shell is three, so the electronic configuration of valence shell for the given element is 3s2, 3p5. Third period starts from atomic number, Z =11 and end at Z = 18. Hence, the atomic number of the given element is 10 + 7 = 17.
Q.3.7: Which element do you think would have been named by
(i) Lawrence Berkeley Laboratory
(ii) Seaborg’s group?
Ans: (i) Lawrencium (Z = 103) and Berkelium (Z = 97).
(ii) Seaborgium (Z = 106)
Q.3.8: Why do elements in the same group have similar physical and chemical properties?
Ans: Same group elements have similar electronic configuration therefore, have similar physical and chemical properties.
Q.3.9: What does atomic radius and ionic radius really mean to you?
Ans: Atomic radius: Atomic radius means size of the atom. It can be measured by X-ray or other spectroscopic methods. If the element is a metal, then the atomic radius is called metallic radius, and if the element is a nonmetal, then it is called covalent radius.
Metallic radius is calculated as half of the internuclear distance separating the metal cores in the metallic crystal. For example, the internuclear distance between two adjacent copper atoms in solid copper is 256 pm Thus, the metallic radius of copper is taken as $\frac{256}{2}$ pm = 128pm.
Covalent radius is defined as one half the distance between the nuclei of two covalently bonded atoms of the same element in a molecule. For example, the bond distance in the Cl2 molecule is 198 pm and half of this distance, 99 pm is taken as the atomic radius of chlorine.
Ionic radius Ionic radius means size of the ion (cation or anion). It can be estimated by measuring the distances between cations and anions in ionic crystal.
A cation is always smaller than its parent atom because effective nuclear charge increases as a result of loss of one or more electrons.
For example, the atomic radius of sodium (186 pm) is larger than ionic radius of Na+ ion (95 pm).
An anion is always larger than its parent atom because effective nuclear charge decreases as a result of addition of one or more electrons.
For example; ionic radius of fluoride ion (F– = 136 pm) is greater than atomic radius of fluorine atom (F = 72 pm).
Q.3.10: How do atomic radius vary in a period and in a group? How do you explain the variation?
Ans: Atomic size decreases as we move from left to right in a period. It is because within a period the electrons enter in the same valence shell and the effective nuclear charge increases with increase in atomic number. As a result of this increased effective nuclear charge, the attraction of the nucleus for outer electrons increases and hence, the atomic size decreases.
Within a group atomic size of the elements increases regularly with increase in atomic number. As we move down a group, number of shells increases by one and the valence shell electrons are farther and farther away from the nucleus. Due to this, nuclear attraction for valence electrons decreases and hence, the size increases.
Q.3.11: What do you understand by isoelectronic species? Name a species that will be isoelectronic with each of the following atoms or ions.
(i) F–
(ii) Ar
(iii) Mg2+
(iv) Rb+
Ans: Isoelectronic species : These have the same number of electrons but different nuclear charges. In case of isoelectronic species as the nuclear charge increases, their size decreases.
(i) F– has 10 electrons (9 + 1).
N3- (7+3),O2-(8+2), Ne(10), Na+(11-1) and Al3+(13 – 3) are some species which are isoelectronic with F– and Mg2+.
(ii) Ar has 18 electrons. Therefore, P3-, S2-, Cl–, K+ and Ca2+ are some species which are isoelectronic to Ar.
(iii) Mg2+ has 10 electrons (12-2) and
(iv) Rb+ has 36 electrons (37-1).Therefore, Br–, Kr and Sr2+ are isoelectronic with Rb+.
Q.3.12: Consider the following species : N3–, O2–, F–, Na+, Mg2+ and Al3+
(a) What is common in them?
(b) Arrange them in the order of increasing ionic radii.
Ans: (i) All the given species have same number of electrons (10e–).
Therefore, all are isoelectronic.
(ii) The ionic radii of isoelectronic species decrease with increase in atomic number (as magnitude of the nuclear charge increases with increase in atomic number). Therefore, their ionic radii increase in the order
Isoelectronic ions = Al3+ < Mg2+ < Na– < F– < O2- < N3-
Atomic number, Z = 13 12 11 9 8 7
Q.3.13: Explain why cation are smaller and anions larger in radii than their parent atoms?
Ans: Cations are always smaller in radii than their parent atoms because by the loss of one or two electrons effective nuclear charge increases. Due to this force of attraction of nucleus for electrons increases and hence, ionic radii decrease.
On the other hand, anions always larger in radii than their parent atoms because by the addition of one or two electrons effective nuclear charge decreases. Due to this, forces of attraction between nucleus and valence shell electrons decreases and hence, ionic radii of anion increase.
Q.3.14: What is the significance of the terms — ‘isolated gaseous atom’ and ‘ground state’ while defining the ionization enthalpy and electron gain enthalpy? (Hint : Requirements for comparison purposes.)
Ans: (i) Ionization enthalpy : It is the minimum amount of energy required to remove an electron from an isolated gaseous atom in its ground state.
A(g) →A+(g) + e–
The force by which an electron is attracted by nucleus is also affected by the presence of other atoms within its molecule or in the neighbourhood. Therefore, ionization enthalpy is determined in gaseous state because in gaseous state interatomic distances are larger and interatomic forces of attractions are minimum. Furthermore, ionization enthalpy is determined at a low pressure because it is not possible to isolate a single atom but interatomic attractions can be further reduced by reducing pressure. Due to these reasons, the term isolated gaseous atom in ground state has been included in definition of ionization enthalpy.
(ii) Electron gain enthalpy : It is the energy released when an isolated gaseous atom in ground state gains an electron to form gaseous anion.
B(g) + e–→ B–(g)
The most stable state of an atom is ground state. If isolated gaseous atom is in excited state, comparatively lesser energy will be released on addition of an electron. So, electron gain enthalpies of gaseous atoms must be determined in their ground states.
Therefore, for comparison purposes, the electron gain enthalpies of gaseous atoms must be determined in their respective most stable state, i.e., ground state.
Q.3.15: Energy of an electron in the ground state of the hydrogen atom is –2.18×10–18J. Calculate the ionization enthalpy of atomic hydrogen in terms of J mol–1.
Ans: Ionization energy is the amount of energy required to remove the electron from the ground state (E1) to infinity (E∞).
E1 = – 2.18 x 10-18 J
E∞ = 0 (Energy of an electron at infinity = 0)
∵ ΔE = E∞ – E1
ΔE = 0 – (-2.18 x10-18 J)
ΔE = 2.18 x 10-18 J
Ionization enthalpy per hydrogen atom = 2.18 x10-18 J
Ionization enthalpy per mole of hydrogen atoms
= 2.18 x 10-18 x 6.022 x 1023 J mol-1
= 13.12 x 105 J mol-1
Hence, the ionization enthalpy of atomic hydrogen in terms of J mol–1 is 13.12 x 105 J mol-1.
Q.3.16: Among the second period elements the actual ionization enthalpies are in the order Li < B < Be < C < O < N < F < Ne. Explain why
(ii) O has lower ΔiH than N and F
7N =1s2, 2s2, $2p_{x}^{1},2p_{y}^{1},2p_{z}^{1}$ (Half-filled configuration)
8O =1s2, 2s2, $2p_{x}^{2},2p_{y}^{1},2p_{z}^{1}$ (Incomplete filled configuration)
9F =1s2, 2s2, $2p_{x}^{2},2p_{y}^{2},2p_{z}^{1}$ (Higher nuclear charge)
Across a period, ionization enthalpy increases as we move from left to right due to decrease in atomic size. But ΔiH of nitrogen is greater than oxygen because N has the more stable electronic configuration (Half filled ), so it is difficult to remove an electron from N than from O.
But in case of F, its higher nuclear charge (+9), the first ionization enthalpy of F is higher than that of O. That’s why O has lower ionization enthalpy than N and F.
Q.3.17: How would you explain the fact that the first ionization enthalpy of sodium is lower than that of magnesium but its second ionization enthalpy is higher than that of magnesium?
Ans: The electronic configuration of Na and Mg are:
Na = 1s2,2s2,2p6, 3s1
Mg = 1s2, 2s2,2p6, 3s2
First ionization enthalpy of sodium (Na = 1s2,2s2,2p6, 3s1) is lower than that of magnesium (Mg = 1s2, 2s2,2p6, 3s2) because the electron to be removed in both the cases is from 3s-orbital but the nuclear charge is lower in Na than that of Mg.
Atomic Size ∝ $\frac{1}{IE}$
After the removal of first electron Na+ acquires inert gas (Ne) configuration (Na+ = Is2, 2s2, 2p6) and hence, removal of second electron from sodium is difficult.
While in case of Mg, after the removal of first electron, the electronic configuration of Mg+ is 1s2, 2s2, 2p6,3s1. In this case 3s1 electron is easy to remove in comparison to remove an electron from inert gas configuration. Therefore, IE2 of Na is higher than that of Mg.
Remember: The species having exactly half- filled or fully filled orbitals have extra ordinarily high ionization enthalpies.
Q.3.18: What are the various factors due to which the ionization enthalpy of the main group elements tends to decrease down a group?
Ans: The ionization enthalpy of the main group elements decreases regularly on moving down the group due to the following two factors:
(i) Atomic size: On moving down the group, atomic size increases due to addition of new orbit. As a result of this forces of attraction of nucleus for valence electrons decreases and ionization enthalpy also decreases.
(ii) Screening effect: On moving down the group, screening effect or shielding effect increases. This effect tends to increase the size of atom. Due to this ionization enthalpy decreases (because forces of attraction between nucleus and electrons decreases).
Q.3.19: The first ionization enthalpy values (in kJ mol–1) of group 13 elements are :
![]()
How would you explain this deviation from the general trend ?
Ans: In general, on moving down the group (13th group) from B to Al, the ionization enthalpy decreases with increase in atomic size and screening effect as expected.
But IE1, of Ga is slightly higher (only 2 kJ mol-1) than IE2, of Al. It is due to poor shielding of the valence electrons by 3d-electrons.
As a result of this effective nuclear charge in Ga is slightly more than that of Al. That’s why (IE1) ΔiH1 of Ga is slightly more than that of Al.
On moving from In to Tl, ΔiH1 of Tl is larger than that of In. It is due to the fact that effective nuclear charge is greater than the shielding effect of all the electrons present in 4f and 5d-electrons.
Q.3.20: Which of the following pairs of elements would have a more negative electron gain enthalpy?
(i) O or F
(ii) F or Cl
Ans: (i) Oxygen and fluorine : Both belong to second period. Electron gain enthalpy generally becomes more negative across a period as we move from left to right.
On moving from O to F, the effective nuclear charge increases and atomic size decreases with increase in atomic number. Due to this, forces of attraction of the nucleus increases for the incoming electron. That’s why electron gain enthalpy becomes more negative for F than that of O.
Furthermore, gain of one electron by F gives F– ion which has stable inert gas configuration. Therefore, the energy released is much higher in going from F to F– than in going from O to O–.
Therefore, electron gain enthalpy of F is much more negative ( -328 kJ mol-1) than that of oxygen (-141 kJ mol-1).
(ii) Within a group, electron gain enthalpy becomes less negative down a group. But electron gain enthalpy of chlorine is more negative (-349 kJ mol-1) than that of the fluorine (- 328 kJ mol-1).This is due to small size of F as the electron-electron repulsions in relatively greater in 2p-orbital than that in the larger 3p-orbital and hence, the incoming electron feels greater repulsion in F than in the Cl. That’s why electron gain enthalpy of Cl is more negative than that of F.
Q.3.21: Would you expect the second electron gain enthalpy of O as positive, more negative or less negative than the first? Justify your answer.
Ans: When an electron is added to oxygen atom to form O– ion, energy is released. Hence, first electron gain enthalpy of oxygen is negative.
O(g) + e–(g) → O– (g); ∆egH = -141 kJ mol–
But when another electron is added to O– ion to form O2- ion, it feels stronger electrostatic repulsion. Hence, addition of second electron takes place with absorption of energy.
O–(g) + e– (g) → O2- (g); ∆egH = +780 kJ mol–
That’s why the second electron gain enthalpy of oxygen is positive.
Q.3.22: What is the basic difference between the terms electron gain enthalpy and electronegativity?
Ans: Electron gain enthalpy is the tendency of an isolated gaseous atom to accept an extra electron to form a gaseous anion while electronegativity is the tendency of an atom of an element to attract the shared pair of electrons towards itself in a covalent bond. Unlike electron gain enthalpy, electronegativity is not a measurable quantity
Q.3.23: How would you react to the statement that the electronegativity of N on Pauling scale is 3.0 in all the nitrogen compounds?
Ans: The statement that electronegativity of N on Pauling scale is 3.0 in all the nitrogen compounds is wrong because electronegativity of any given element is not constant. Electronegativity varies depending on the element to which it is bound. It increases as the oxidation state of the element increases or percentage of s-character of hybrid orbital increases.
For example, EN of N in NO2 (oxidation state +4) is higher than N in NO where oxidation state is +2
Q.3.24: Describe the theory associated with the radius of an atom as it
(a) gains an electron
(b) loses an electron
Ans: (a) When a neutral atom gains an electron to form anion, its radius increases. In an anion per electron nuclear forces decreases due to increase in number of electrons and as a result of decrease in effective nuclear charge, radius of an anion increases.
For example, ionic radius of Cl– ion is greater than the radius of its parent atom Cl.
(b) When an atom loses an electron to form cation, its radius decreases. In a cation per electron nuclear forces increases due to decrease in number of electrons. As a result of this, effective nuclear charge increases and the radius of cation decreases. For example, ionic radius of Na+ is smaller than the radius of its parent atom Na.
3.25: Would you expect the first ionization enthalpies for two isotopes of the same element to be the same or different? Justify your answer.
Ans: Isotopes of an element have same number of electrons, electronic configuration and thus, the same nuclear charge. Also, the ionization enthalpy depends upon the electronic configuration and effective nuclear charge. Hence, first ionization enthalpies of two isotopes of the same element are expected to be same.
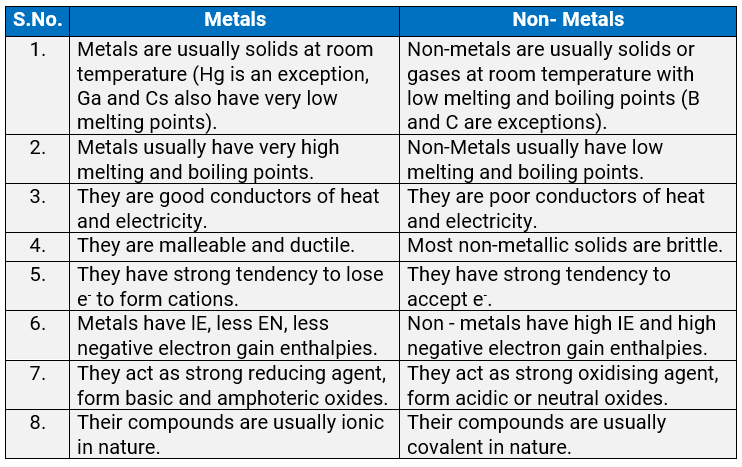
3.26: What are the major differences between metals and non-metals?
Ans:
3.27: Use the periodic table to answer the following questions.
(a) Identify an element with five electrons in the outer subshell.
(b) Identify an element that would tend to lose two electrons.
(c) Identify an element that would tend to gain two electrons.
(d) Identify the group having metal, non-metal, liquid as well as gas at the room temperature.
Ans: (a) General electronic configuration of elements having five electrons in the outer sub shell is ns2 np5. This configuration belongs to halogen family, i.e., F, Cl, Br, I, At.
(b) Elements of second group are known as alkaline earth metals (Mg, Ca, Sr, Ba, etc). Their general electronic configuration for valence shell is ns2. These elements form dipositive cations by the lose of two electrons easily.
(c) 16th group elements such as O, S, Se, etc., have a tendency to accept two electrons because by the gain of two electrons they attain noble gas configuration. Their general electronic configuration for valence shell is ns2 np4.
(d) Group 1 or 17 of the Periodic Table contains metal, non-metal, liquid as well as gas at the room temperature, e.g., H2 is a non-metal and in gaseous state at room temperature. All other elements of this group are metals. Cs is a liquid metal. similarly, Br2 is a liquid non-metal while other elements of this group are gaseous non-metals. Iodine can form I+ so it has somewhat metallic properties.
Q.3.28: The increasing order of reactivity among group 1 elements is Li < Na < K < Rb CI > Br > I. Explain.
Ans: The elements of Group – I have only one e– in their respective valence shells and thus have a strong tendency to lose this e– to form cation.
The tendency to lose an electron depends upon the ionization enthalpy (IE).
The IE decreases down the group. Hence, the reactivity increases down the group (Li < Na < K < Rb < Cs).
On the other hand, the elements of group 17 have seven electrons in their respective valence shells and thus have strong tendency to accept one more electron to make stable configuration. So tendency to accept electrons depends on electron gain enthalpy and electronegativity. Since both of them decreases down the group, the reactivity therefore decreases (F > Cl > Br > I).
Q.3.29: Write the general outer electronic configuration of s-, p-, d- and f- block elements.
Ans: s-block elements – ns1-2 (n = 2- 7)
p-block elements- ns2np1 – 6 (n = 2- 7)
d-block elements- (n -1) d1-10 ns 0-2 (n = 3- 7)
f-block elements- (n-2) f1-14 (n -1) d0-1 ns2 (n = 6-7)
Q.3.30: Assign the position of the element having outer electronic configuration
(i) ns2np4 for n=3
(ii) (n-1)d2ns2 for n=4, and
(iii) (n-2)f7 (n-1) d1 ns2 for n=6, in the periodic table.
Ans: (i) ns2np4 for n = 3
n = 3 means element belongs to third period. Since, last electron enters in the p-orbital, it belongs to p-block.
For p-block elements,
Group number =10 + valence shell electrons
= 10 + (2 + 4) = 16
Hence, the element belongs to 16th group.
The electronic configuration = 1s2, 2s2, 2p6, 3s2, 3p4
Thus, the element is Sulphur (16S).
(ii) (n -1) d2 ns2 for n = 4
n=4 means the element belongs to 4th period. Since, last e– enters in d-orbital, the given element belongs to d-block.
For d-block elements,
Group number = no. of d – e– + no. of ns – e–
= 2 + 2 = 4
Hence, the element belongs to 4th group.
The complete electronic = 1s2, 2s2, 2p6, 3s2, 3p6, 3d2, 4s2
Thus, the element is titanium(22Ti).
(iii) (n -2)f7(n-1)d1ns2 for n = 6
n = 6 means, the element belongs to 6th period. Since, last e– enters in f-orbital, the given element belongs to f-block and all f-block elements are the members of 3rd group.
Hence, the element belongs to 3rd group.
The complete configuration of the element is as follows,
1s2, 2s2, 2p6, 3s2, 3p6, 3d10, 4s2, 4p6, 4d10, 5s2, 5p6, 4f7, 5d1 6s2
Thus, the element is Gadolinium (Gd).
Q.3.31: The first (ΔiH1) and the second (ΔiH2) ionization enthalpies (in kJ mol–1) and the (ΔegH) electron gain enthalpy (in kJ mol–1) of a few elements are given below:
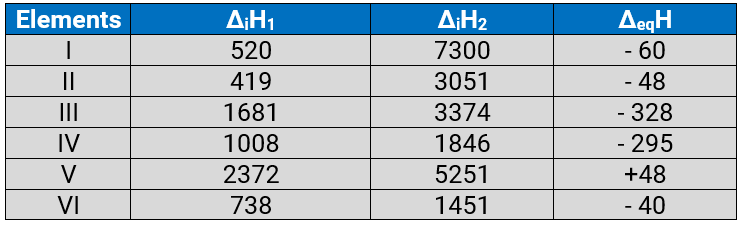
Ans: (a) The least reactive element is element (V) because it has highest Δ1H1 (first ionization enthalpy) and positive electron gain enthalpy (ΔegH). The element (V) is an inert gas because inert gases have positive electron gain enthalpy.
(b) The most reactive metal is element (II) because it has the least ΔiH1(the first ionization enthalpy) and low negative electron gain enthalpy (ΔegH).
(c) The most reactive non-metal is element (III) because it has high ΔiH1 (first ionization enthalpy) and a very high negative electron gain enthalpy (ΔegH).
(d) The least reactive non-metal is element IV. The element IV has not so high ΔiH1 but it has high negative electron gain enthalpy (ΔegH).
(e) The metal which can form a stable binary halide of the formula MX2 is element (VI). Element VI is alkaline earth metal because it has low ΔiH1 but higher than that of alkali metals. Further, it has lowest second ΔiH2 value.
(f) Element I have low ΔiH1 but a very high ΔiH2. It has less negative electron gain enthalpy. So, it can form predominantly stable covalent halide of the formula MX.
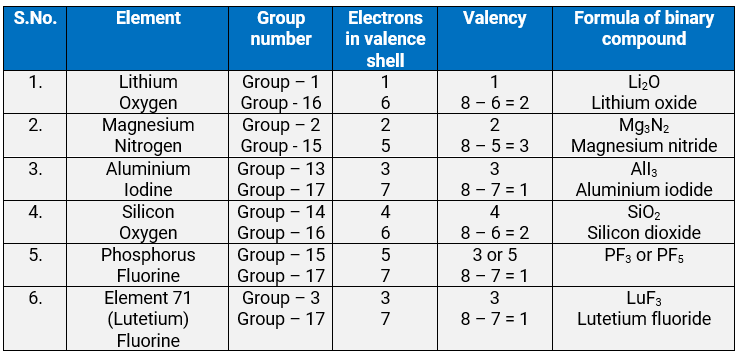
Q.3.32: Predict the formulas of the stable binary compounds that would be formed by the combination of the following pairs of elements.
(a) Lithium and oxygen
(b) Magnesium and nitrogen
(c) Aluminium and iodine
(d) Silicon and oxygen
(e) Phosphorus and fluorine
(f) Element 71 and fluorine
Ans:
Q.3.33: In the modern periodic table, the period indicates the value of:
(a) Atomic number
(b) Atomic mass
(c) Principal quantum number
(d) Azimuthal quantum number.
Ans: Correct option (c),
Explanation: In the modem Periodic Table, the period indicates the value of principal quantum number. Hence, the correct option (c).
Q.3.34: Which of the following statements related to the modern periodic table is incorrect?
(a) The p-block has 6 columns, because a maximum of 6 electrons can occupy all the orbitals in a p-shell.
(b) The d-block has 8 columns, because a maximum of 8 electrons can occupy all the orbitals in a d-subshell.
(c) Each block contains a number of columns equal to the number of electrons that can occupy that subshell.
(d) The block indicates value of azimuthal quantum number (l ) for the last subshell that received electrons in building up the electronic configuration.
Ans: Statement (b) is incorrect.
Explanation: The correct statement (b) is; the d-block has 10 columns, because a maximum of 10 electrons can occupy all the orbitals in a d-subshell. All other given statements are correct.
Q.3.35: Anything that influences the valence electrons will affect the chemistry of the element. Which one of the following factors does not affect the valence shell?
(a) Valence principal quantum number (n)
(b) Nuclear charge (Z )
(c) Nuclear mass
(d) Number of core electrons.
Ans: Correct answer (c)
Explanation: Nuclear mass (protons + neutrons) does not affect the valence shell, only protons, i.e., nuclear charge affects the valence shell.
Q.3.36: The size of isoelectronic species — F–, Ne and Na+ is affected by
(a) nuclear charge (Z )
(b) valence principal quantum number (n)
(c) electron-electron interaction in the outer orbitals
(d) none of the factors because their size is the same.
Ans: Correct statement (a),
Explanation:
The size of isoelectronic species; F–, Ne and Na+ is affected by nuclear charge (Z). With increase in nuclear charge (atomic number), the size of the isoelectronic species decreases.
For example, F– (+ 9) > Ne (+10) > Na+(+11). Therefore, statement (a) is correct while all other statements are wrong.
Q.3.37: Which one of the following statements is incorrect in relation to ionization enthalpy?
(a) Ionization enthalpy increases for each successive electron.
(b) The greatest increase in ionization enthalpy is experienced on removal of electron from core noble gas configuration.
(c) End of valence electrons is marked by a big jump in ionization enthalpy.
(d) Removal of electron from orbitals bearing lower n value is easier than from orbital having higher n value.
Ans: Incorrect statement (d)
The correct statement is removal of electron from orbitals bearing lower n value is difficult than from orbital having higher n value, due to decrease in effective nuclear charge with increase in the value of n.
Q.3.38: Considering the elements B, Al, Mg, and K, the correct order of their metallic character is :
(a) B > Al > Mg > K
(b) Al > Mg > B > K
(c) Mg > Al > K > B
(d) K > Mg > Al > B
Ans: Option (d)
Explanation: In a group, metallic character increases from top to bottom as ionisation energy decreases. Therefore, metallic character of Al, is greater than B i.e., Al > B.
In a period, metallic character decreases from left to right as tendency to lose electron decreases. Therefore, metallic character of Al, Mg and K decrease in order: K > Mg > Al. Therefore, the correct order is K > Mg > Al > B i.e., Option (d).
Q.3.39: Considering the elements B, C, N, F, and Si, the correct order of their non-metallic character is :
(a) B > C > Si > N > F
(b) Si > C > B > N > F
(c) F > N > C > B > Si
(d) F > N > C > Si > B
Ans: Options (c)
Explanation: Non-metallic character in a group decreases from top to bottom thus C is more non- metallic than Si.
but non-metallic character increases in a period when we move from left to right. This is because ionisation energy increases in a period and decreases in group. Therefore, among B, C, N, F and Si, the non-metallic character decreases in the order F > N > C > B > Si i.e., option (c) is correct.
Q.3.40: Considering the elements F, Cl, O and N, the correct order of their chemical reactivity in terms of oxidizing property is:
(a) F > Cl > O > N
(b) F > O > Cl > N
(c) Cl > F > O > N
(d) O > F > N > Cl
Ans: Option (b),
Explanation: In a group oxidising power (i.e., tendency to gain electron) decreases from top to bottom as the size increases. Thus, F is a stronger oxidizing agent than Cl.
When we move left to right in a period, Oxidizing power increases because size decreases. Therefore, overall decreasing power of oxidizing power is: F > O > Cl > N i.e., option (b) is correct.



